Alzheimer's disease
exploring the mechanisms of functional connectivity changes in Alzheimer’s.
Understanding Functional Connectivity in Neurodegeneration
Alzheimer’s Disease (AD) is a progressive neurodegenerative disorder characterized not only by cognitive decline but also by widespread disruptions in brain network dynamics. Functional connectivity—how different brain regions communicate—evolves throughout the disease continuum, from preclinical stages to severe dementia. Understanding these changes is key to developing early biomarkers and intervention strategies.
From Connectivity to Mechanisms: Computational Modeling of AD
The human brain is a complex network, and its dysfunction in AD cannot be fully understood through isolated observations. Computational models allow us to explore how neurobiological alterations—such as synaptic dysfunction, neuronal loss, and network disconnection—contribute to the progressive breakdown of functional connectivity. These models provide a framework for linking neuroimaging findings with underlying neural mechanisms, offering a deeper insight into disease progression.
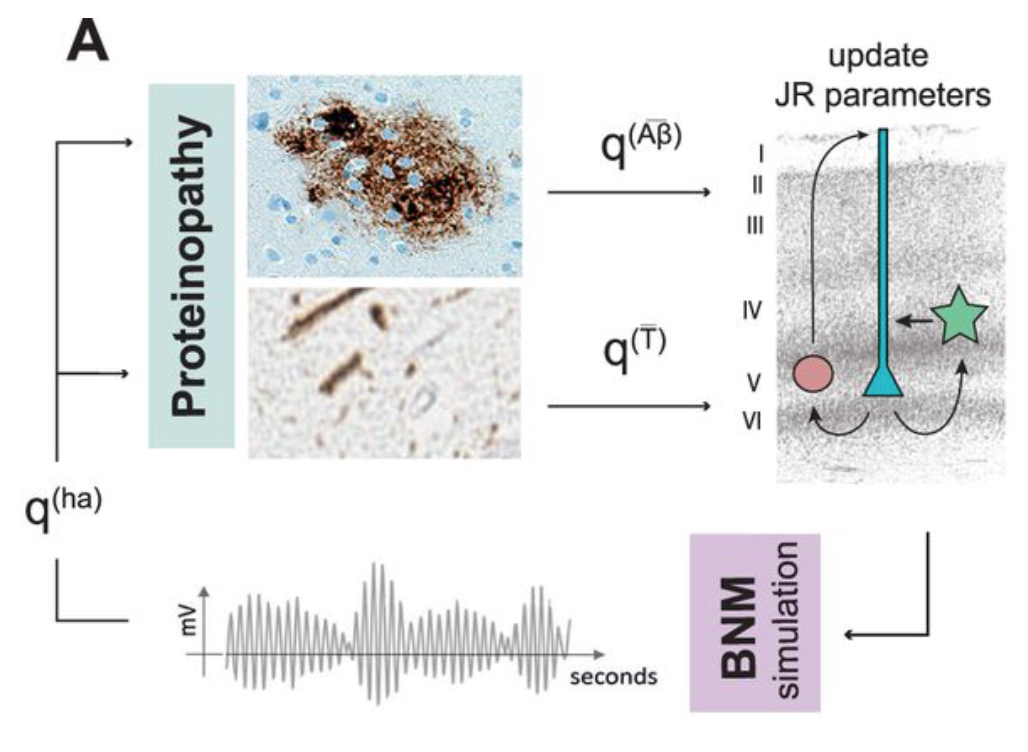
The Role of Proteinopathy in Brain Dynamics
A hallmark of AD is the accumulation of pathological proteins, such as beta-amyloid and tau, which spread across the brain in characteristic patterns. But how do these molecular changes translate into altered neuronal activity and network function? By integrating proteinopathy maps with computational modeling, we can investigate how these pathological processes shape functional connectivity and contribute to cognitive impairment.
Related citations (Cabrera-Álvarez et al., 2024)
Bridging Scales: From Neurons to Whole-Brain Networks
Alzheimer’s research requires a multiscale perspective, connecting microscopic cellular pathology with macroscopic brain dynamics. Cutting-edge techniques—ranging from large-scale brain simulations to multimodal neuroimaging—are helping to unravel how local molecular events cascade into system-wide dysfunction. This knowledge is essential for designing targeted interventions that go beyond symptom relief and tackle the fundamental mechanisms of neurodegeneration.
Future Directions: Towards Personalized Models of AD Progression
While group-level studies have provided valuable insights into AD progression, there is a growing need for personalized approaches. Could we predict an individual’s disease trajectory based on their unique brain network properties? How can computational models be leveraged to refine therapeutic strategies? The future of AD research lies in combining neuroimaging, computational modeling, and artificial intelligence to develop individualized disease predictions and interventions.
Non-Invasive Brain Stimulation (NIBS) as a Potential Therapeutic Approach
Given the widespread network dysfunction in AD, non-invasive brain stimulation (NIBS) techniques, such as transcranial magnetic stimulation (TMS) and transcranial alternating current stimulation (tDCS), have emerged as potential tools to restore normal functional connectivity patterns. These techniques modulate cortical excitability and network dynamics, potentially counteracting the disruptions observed in AD. By integrating NIBS with computational models of brain activity, we can refine stimulation protocols to target specific network alterations and explore their efficacy in slowing cognitive decline.